Clinical Evaluation of the ProSonic™ System
ProSonic™‘s ultrasonic fastwire® technology is currently being evaluated in a multi-centre clinical study following first-in-human use. The primary focus of this evaluation is understanding device behaviour during the crossing of complex peripheral chronic total occlusions (CTOs).
As part of this work, additional investigation is underway to better understand the behaviour of radial and axial ultrasonic energy during crossing of occlusive disease. These exploratory observations are intended to inform future clinical and regulatory strategy.
FREEFLOW Study Overview
Study objective
The FREEFLOW (FastWire REvascularisation of Extremities, For Lower Limbs) study is evaluating the safety and efficacy of the ProSonic™ System to traverse peripheral chronic total occlusions, with a primary focus on facilitating crossing of the CTOs and enabling intraluminal placement of follow-on devices beyond the CTO.
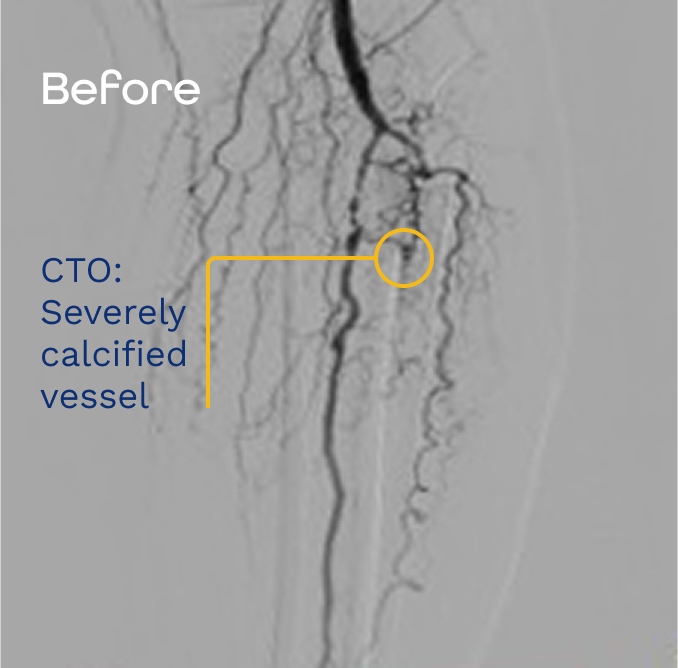
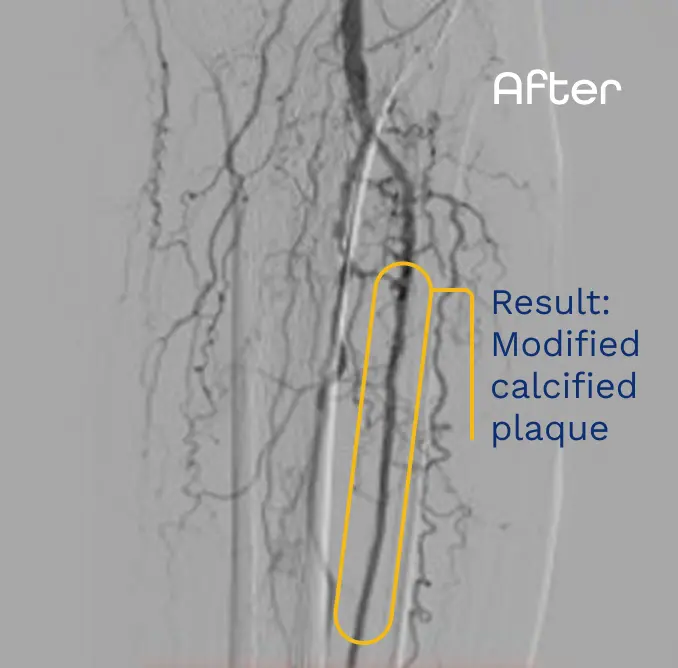
Presented NCVH 2023 by Dr. Craig Walker. Images are for informational purposes only. No claims of clinical benefit are made.
Study Measures
Safety
Efficacy in crossing
Other performance attributes
Data generated from the study are intended to inform future regulatory submissions and staged regulatory engagement.
Scope of Clinical Investigation
The current clinical programme is designed to prioritise understanding of crossing behaviour in complex peripheral CTOs.
Insights from this work will guide future clinical development and regulatory planning as evidence evolves.
Regulatory Status
The ProSonic™ System’s FastWire® technology is currently being investigated through an IDE and is not approved/cleared for sale or commercial use in any market.
Collaboration & Clinical Development
Versono Medical is actively engaged with clinicians, research groups and centres, and industry stakeholders to advance clinical understanding of CLTI and explore future development pathways.
Connect With Versono
Versono is developing an intravascular ultrasonic platform focused first on crossing complex peripheral chronic total occlusions, using a distinct mechanism of action based on proprietary axial and radial ultrasonic energy delivery.
Application development is evidence-led and phased, with an initial focus on crossing and a longer-term platform vision informed by clinical need.
We are now welcoming investor interest as the company enters its next stage of development.